Idiopathic pulmonary fibrosis (IPF) is a chronic, progressive form of lung disease characterized by fibrosis or scarring of the supporting framework of the lungs. In the United States, IPF affects between 132,000 and 200,000 people. Approximately 50,000 new cases are diagnosed each year, and as many as 40,000 Americans die from IPF each year. Nearly 50 percent of IPF sufferers die within three years of diagnosis. There is no cure or FDA-approved treatments for IPF in the United States and lung transplantation remains the most viable course of treatment to extend the lives of those with IPF.
We believe that our drug candidates may play a role in the treatment of lung fibrosis. Preclinical studies have been conducted and revealed that treatment with GR-MD-02 showed a robust effect in reducing lung fibrosis.
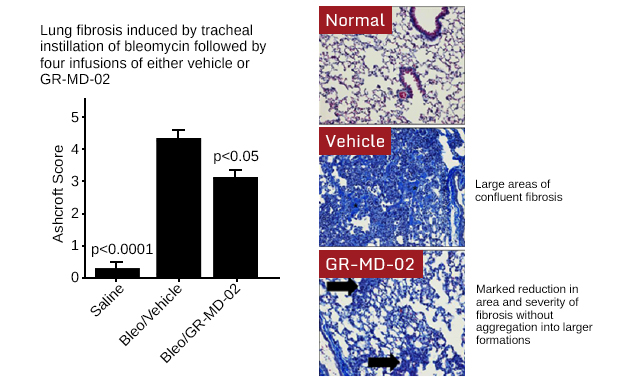
In preclinical studies, lung fibrosis was induced in mice by the intra-tracheal instillation of bleomycin, a standard model in the pharmaceutical industry for simulating the human disease and testing potential therapeutic agents. After induction of lung fibrosis therapy, treatment with Galectin Therapeutics’ two anti-galectin drugs, GM-CT-01 and GR-MD-02, was started either immediately (prevention study) after or ten days after (treatment study) instillation of bleomycin.
In the prevention study, both drugs markedly reduced lung weight and hydroxyproline content, with reduction of histological evidence of inflammation and fibrosis when compared to vehicle-treated bleomycin mice. In the treatment study, GR-MD-02 was more effective than GM-CT-01. These results suggest that further studies are warranted to evaluate taking GR-MD-02 into development for the indication of IPF.
The two preclinical studies were performed in collaboration with Biomodels in Watertown, Mass., which has extensive experience with this mouse model of lung fibrosis.